Explain the Mechanism of Photosynthesis.
Understanding the Mechanism of Photosynthesis:
Evidence for the Existence of Light and Dark Reactions: F.F. Blackman, a British plant physiologist who discovered that photosynthesis is a two-step process (1905), only one of which uses light directly.
He cultivated plants under different but controlled carbon dioxide concentrations, different light intensities and different temperatures and noted the effects of these variables on the rate of photosynthesis.
Under low light intensity, photosynthesis is enhanced by increasing light, but is unaffected by increases in temperature and carbon dioxide.
Also read | Objective, Importance and Limitations of Animal Breeding.
When light intensity is high, increases in both temperature and carbon dioxide accelerate photosynthesis. Black concluded that the initial “light” reactions are independent of temperature, while the second “dark” reactions are independent of light yet are limited by carbon dioxide and controlled by enzymes.
By estimating Q10 ≈ 2.5 of the photosynthetic reaction, he studied the process under optimal conditions, especially of adequate light and supply of carbon dioxide.
The Q10 value provides an idea about the complexity of the reactions. For physical process the Q10 value ranges from 1.2 to 1.4 and for chemical reactions . it is generally 2 to 3 and for photo-chemical reaction, it is 1.
Also read | The Mechanism of Stomatal Opening.
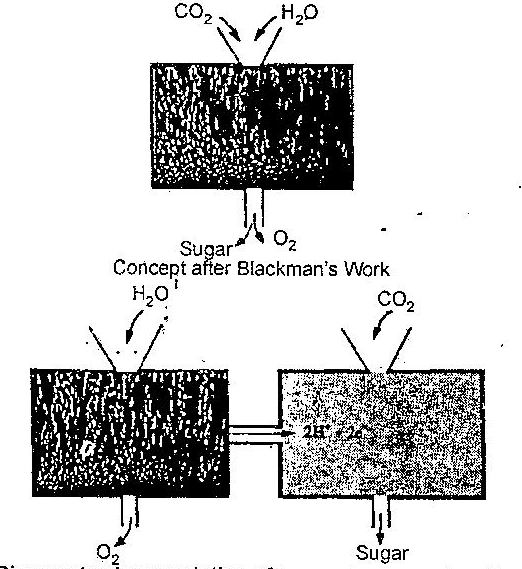
Diagram to show evolution of concepts concerning the mechanism of photosynthesis. The box above represents the concept at the end of the last century.
Also read | Educational Excursion.
After Blackman conducted the experiments to determine the Q10, of the photosynthetic reaction, it was concluded that the reactions in the ‘black magic box’ consisted at least of two sub-sets of reactions, the light and the dark reactions.
The actual roles, that in the light box there is photolysis of water and in text dark box there is reduction of carbon dioxide was established much later.
In the above figure, there are two boxes out of which one is light containing the chlorophyll pigments on which light shone and which represented the light reactions, and the other is a “dark” box representing the dark reactions.
The German Nobel laureate, Otto Warburg, and his American pupils, Robert Emerson and William Arnold performed a series of experiments with alternating flashes of light and darkness given to the unicellular green alga, Chlorella.
Also read | List of physical hazards during early childhood period.
They obtained intermittent light and darkness with the help of a mechanical device, consisting of an electric motor to which was attached a rotating disc with sectors cut in different sizes was set in the light path.
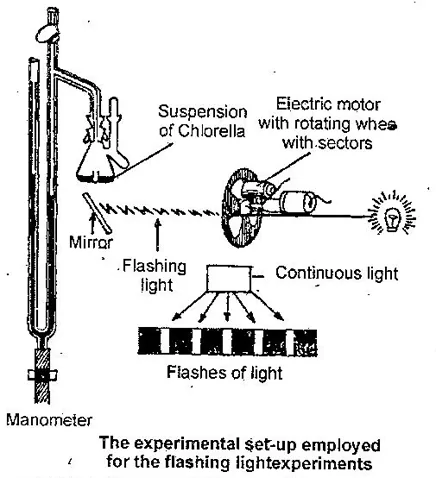
It is found that on separating light and dark periods by appropriate intervals, the oxygen yield per unit light shone increased, by as much as 400% over the control where the same amount of light was given continuously, confirming that photosynthesis consisted of both light and dark reactions.
Also read | Explain Starch Sucrose Inter Conversion.
It was also found later that dark reactions were much slower than light reactions hence the light given continuously was utilized less efficiently.
The Role of Light Reaction.
Photolysis of Water: In the early 1930s, he conducted experiments with sulfur bacteria that undergo a photosynthetic process resembling traditional photosynthesis.
However, these bacteria use hydrogen sulfide instead of water and produce sulfur rather than oxygen. Van Niel learned that in these bacteria, hydrogen from the hydrogen sulfide was combining with carbon dioxide in a two-stage reaction to form a carbohydrate.
When he applied his discovery to the mechanisms of green plants, he theorized that a parallel two-part process was happening in photosynthetic plants, one in which light split water into oxygen and hydrogen, which would combine with carbon dioxide to form a carbohydrate. The reactions associated are:
Also read | Classification of IUB system enzymes categories.
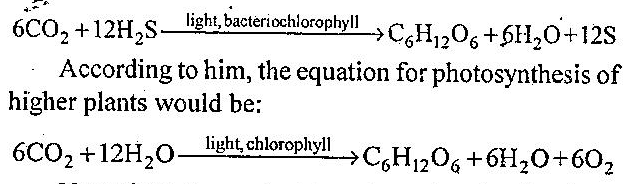
Van Niel’s theory had two important implications. First, his experiments helped demonstrate the fundamental equivalence of photosynthesis in bacteria and green plants.
Second, the assertion that photosynthesis begins with the splitting of water (i.e. photolysis) into hydrogen and oxygen meant that a previously unexpected a separation of charges was occurring during the process.
Also read | Characteristics of Life Span Development.
The role of H2O and H2S can be explained in the following equation. This discovery helped lay the groundwork for modern studies into the mechanisms of photosynthesis, particularly into a complex biochemical process called the electron transfer chain.
H2S → 2H+ + 2e- + S
H2O → 2H+ + 2e- + ½ O2
The confirmation of evolution of O2 from H2O is given by two American chemists, Samuel Ruben and Martin D.Kamen working in U.S. Atomic Energy Commission.
They used ‘heavy water’, H2O18, to yield 18O2 gas, which had showed that the oxygen gas produced in photosynthesis comes from water.
Also read | Osmoregulation in Terrestrial Environment.
Now the photosynthesis is considered as the chemical, process involving a “redox” reaction (i.e. consisting of a reduction and another oxidation event). It involves transfer of hydrogen or electrons from H2O to CO2 and a reductant H2O was oxidized and an acceptor O2 was reduced.
Photoreduction Production of Reducing Power-NADPH
The English biochemist Robin Hill showed that isolated chloroplasts give off oxygen in the presence of unnatural reducing agents like iron oxalate, ferricyanide or benzoquinone after exposure to light. The reaction is now called Hill reaction and it is given below:
Also read | The Structural Variations of Vertebrate Kidney.
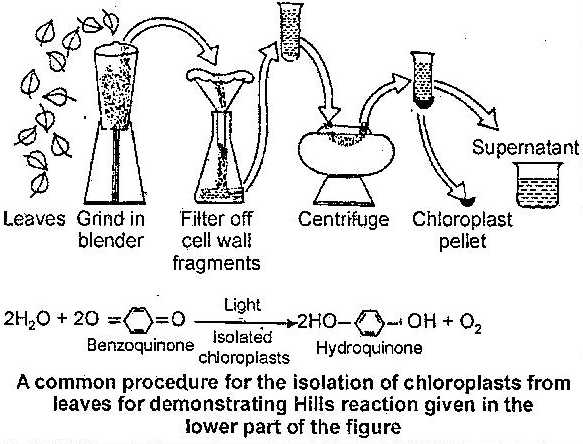
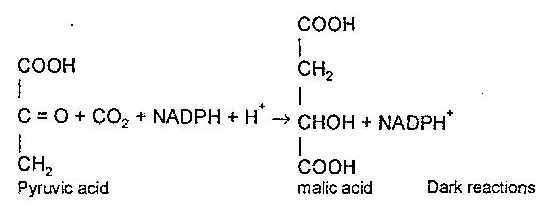
After some time, two American biochemists Wolf Vishniac and Severo Ochoa showed that the photolysis of H2O by illuminated chloroplasts led to the reduction of TPN to TPNH2 (now referred to as NADPH2) along with the CO2 fixation reactions such as carboxylation of pyruvic acid to malic acid. The reactions are shown below.
Also read | The Molecular Organization of Cell Membrane.
H2O → 2H+ + 2e- + ½ O2
NADP + 2H+ → NADP + H+
Photophosphorylation Production of ATP.
The production of ATP using the energy of sunlight is called photophosphorylation. Only two sources of energy are available to living organisms: sunlight and reduction-oxidation (redox) reactions.
Also read | Isoelectric Focusing and Ion Exchange Chromatography.
In photophosphorylation, light energy is used to create a high-energy electron donor, and a lower-energy electron acceptor. Electrons then move spontaneously from donor to acceptor through an electron transport chain.
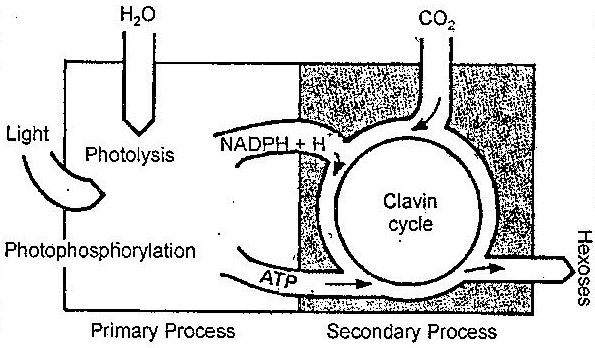
The requirement for ATP in dark reactions of photosynthesis cannot be fulfilled by the ATP supplied by respiration via the ordinary pathway of oxidative phosphorylation. A general scheme for primary and secondary processes is shown in the following figure.