Oxygen Cycle Complete Overview.
Oxygen Cycle is the second most abundant element in the atmosphere and is present to the extent of about 21 per cent and is essential for all life processes.
Atmosphere and oceans are the main pool for oxygen. The oxygen dissolved in water (oceans, etc.) is responsible for sustaining aquatic plants and animals.
It is also a constituent of oxide and carbonate ores. There is a continuous exchange of oxygen between the atmosphere and water surfaces of all water bodies.
Also read | Fungicides and Herbicides.
The total amount of oxygen in the biosphere is constant. The oxygen cycle is based on the exchange of oxygen among the segments of the environment, viz., atmosphere, hydrosphere, lithosphere and biosphere. It plays a key role in atmospheric chemistry and life process.
The different steps involved in oxygen cycle are:
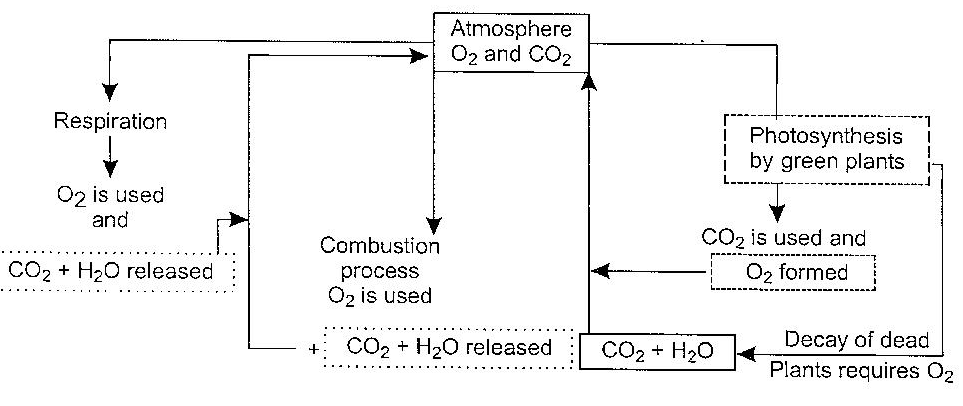
During respiration, organisms including animals and human beings consume oxygen from the air and give carbon dioxide into the atmosphere.
Also read | What is Green House effect?
The green plants utilize carbon dioxide for photosynthesis, in which carbon dioxide reacts with water in presence of solar energy and a catalyst (chlorophyll) to produce carbohydrates and the formed oxygen is released into the atmosphere
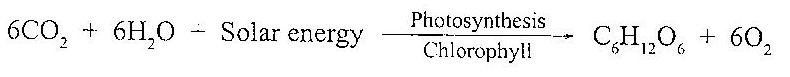
Oxygen is needed by bacteria and fungi during decomposition of organic matter. The products of decomposition (CO2 and H2O) are released into the atmosphere. The released CO2 and H2O are utilised during photosynthesis by green plants.
Also read | Ultraviolet Spectroscopy.
In the upper atmosphere, solar energy splits H2O to give H2 and O2 which are released into the atmosphere

The above processes complete the oxygen cylce. The oxygen content in the atmosphere remains more or less constant. The oxygen cycle is represented as shown in Fig. Below
Also read | Non-Dispersive Infrared Spectroscopy.
Under certain circumstances, there may be imbalance in the oxygen cycle. For example, deforestation reduces the extent of photosynthesis (which is a major source of oxygen in the atmosphere); this leads to reduction of oxygen content in the atmosphere.
This loss, however, is compensated by sulphate reducing bacteria occurring in anaerobic environment; these bacteria use sulphate ions as an oxygen source for oxidizing organic matter.
SO42 + 2C → 2CO2 + S2
Also read | Depletion of Ozone Hole.
The above reaction gives carbon dioxide, which is released into the atmosphere and utilized in photosynthesis leading to the release of oxygen.


